In this exclusive Equine Permaculture series, we have been ‘digging deeper into soils’ and explored what soils are, how soil is formed, the different types of soil and how they sustain life through the soil-food-web.
In Part Three, we described soil sampling and started to investigate some important parameters of soil health using DIY techniques. In this Part Four, we will continue with soil laboratory tests and how to interpret these results.
You can use soil tests as a diagnostic tool or to identify trends through time, which can help you in your decision-making process on how to improve your soils and, ultimately, enhance your pasture health.
Soil laboratory testing
The primary goal of soil testing is to inform efficient and effective resource management on your property. Soil testing is the most accurate way to determine the current state of your soils, its composition, what nutrients it may need (or that are in excess) and other characteristics, such as pH and salinity. Soil testing is also useful for identifying contaminated sites (e.g. elevated levels of heavy metals, pesticides, etc.).
Once you have collected your soil samples (see Part Three), you will have to send these to a soil-testing laboratory to have them accurately analysed.
It is important to recognise the values obtained when a soil sample is analysed are of little use as raw analytical data. In order to make use of the values in predicting nutrient needs, the test must be calibrated.

These calibrations are based on nutrient response research with representative soils (and conditions) – ranging from deficient to adequate for each nutrient of concern. These calibrations are also plant/crop or soil-specific. The sufficiency/optimal range (or typical range, in some cases) is usually provided in the column to the right of your results (for quick comparison).
Soil testing laboratory methods vary, which may influence results and sufficiency ranges. This means if you send the same soil sample to two or three different labs, you may see some variation in the results, as this is largely determined by the analysis techniques and calibrations used at that lab, and may be specific to a region (i.e. state).
Therefore, it is important to check lab techniques/methods used and if the lab is accredited by an authority, such as NATA (National Association of Testing Authorities) or ASPAC (Australian Soil and Plant Analysis Council).
There are other national and international accreditation systems. By visiting the lab’s website, you can find most of this information, as well as their pricing!
Interpreting results
You finally received your soil report of your soil samples, but what does it tell you and how should you interpret these results to make an informed decision?
As mentioned earlier, there are different parameters we can measure in soil and the selection of these listed parameters is determined by the soil analysis packages that you chose when you submitted your samples. Some of these parameters are reasonably simple, such as identifying soil type, which you may already have done yourself by following the instructions outlined in Part Three in the last issue.
When it comes to the nutrients, it is important to note soil tests measure nutrients that are expected to become plant-available, rather than the total amounts of nutrients in soil. Measurements of total nutrient content are not useful indicators of sufficiency for plant growth, because only a small portion of the nutrients in the soil are plant-available.
Roots take up plant-available nutrients as positively (cations) or negatively (anions) charged ions from the soil (see Image B) with the help of structural elements, such as carbon (C), hydrogen (H) and oxygen (O). So, when you see your soil tests results, keep in mind nutrients are expressed as plant-available or extractable/exchangeable (using various test methods, such as Merlich, Morgan, Bray, Olsen, DTPA, etc.). These testing methods will typically be listed in your results as reference.
It is also important to recognise soil test results can be viewed in three categories:
- Low or yes, a fertiliser addition will likely increase growth and productivity;
- High or no, a fertiliser addition will not likely increase growth or yield;
- Intermediate or maybe, a fertiliser addition may increase growth or productivity.
This categorisation is based on the sufficiency/optimal level range, and is often displayed per soil types or can be presented in a bar figure for selected soil type (see Image D on the next page).
Categorisation of soil test results into ‘yes (low), ‘no’ (high) and ‘maybe’ (medium) assists us in understanding the limits and benefits of using soil test results for making nutrient and soil management recommendations. This will be discussed in more detail in the last part of this series in a future issue!
Let’s now look at the different nutrients and other soil characteristics that are listed on your report, and discuss what they tell you. Please note, your results may be expressed in different measuring units, such as parts per million (ppm) or m/kg (which is the same 1:1 for soil).
Soil type
Soil type is reported as the colour and texture of the soil. Both colour and texture are indicators of properties of the soil, and are taken into account when interpreting other soil chemical results, such as Cation Exchange Capacity (CEC).

Soil colour
Soil colour has little direct influence on its chemical, physical or biological attributes, but when considered with other observations, can be very useful.
Often, soils of darker colour are higher in organic matter than lighter coloured soils. Red colour can be related to un-hydrated iron oxides present in well drained soils; yellow or mottled coloured soils may be related to hydrated iron oxides, which may occur where soils are saturated for long periods and/or poorly drained.
The Munsell Soil Colour Charts are internationally accepted as being the standard guide to discern soil colour classification. You can view it online at: https://bit.ly/2HPfNNu.
Soil texture
The texture of a soil is an indication of soil type and its properties. It is always taken into account when interpreting the other results and preparing fertiliser recommendations due to leakage issues. Soil texture is measured separately for Mineral Soils and for Organic Soils; for example, peat.
Soil texture can be measured in two ways, including:
- Field method: Where a small handful of moistened soil is squeezed between the thumb and forefinger to produce a ribbon. The length of the ribbon before breaking and the ‘feel’ of the soil (sandy, silky, etc.) provide an indication of the texture. To improve consistency of results, this test is usually done by experienced laboratory technicians. However, the method remains subjective and the results may differ slightly between assessors. Slight variations are of no real concern to the final fertiliser recommendations.
- Mechanical method: A mechanical sieving process is used to separate and quantify the percentages of sand, silt and clay in a soil. This method is more time-consuming and expensive than the field method, so it is used where greater accuracy is required (e.g. for research).
Organic carbon
Soil organic matter (OM) is a surrogate for soil carbon and is measured as a reflection of overall soil health. Organic matter results from partly decayed plant and animal residues in various degrees of decomposition in the soil.
Soil organic matter assists in maintaining soil structure, and the supply and retention of nutrients, air and water. When monitored for several years, it gives an indication whether soil quality is improving or degrading.
Soil OM is important to a wide variety of soil chemical, physical and biological properties. As soil OM increases, so does CEC, total nitrogen (N) content and other soil properties, such as water-holding capacity and microbiological activity. If a soil is low in organic matter, the soil test will result in a low organic carbon level. Preferred levels are above 2%.
Soil pH
Soil pH is a measure of the alkalinity or acidity of the soil. A pH value of 7 is neutral. Values below 7 are defined as acidic and those above are alkaline. The soil pH can influence the availability of nutrients to plants, and potential toxicity of aluminium and hydrogen.
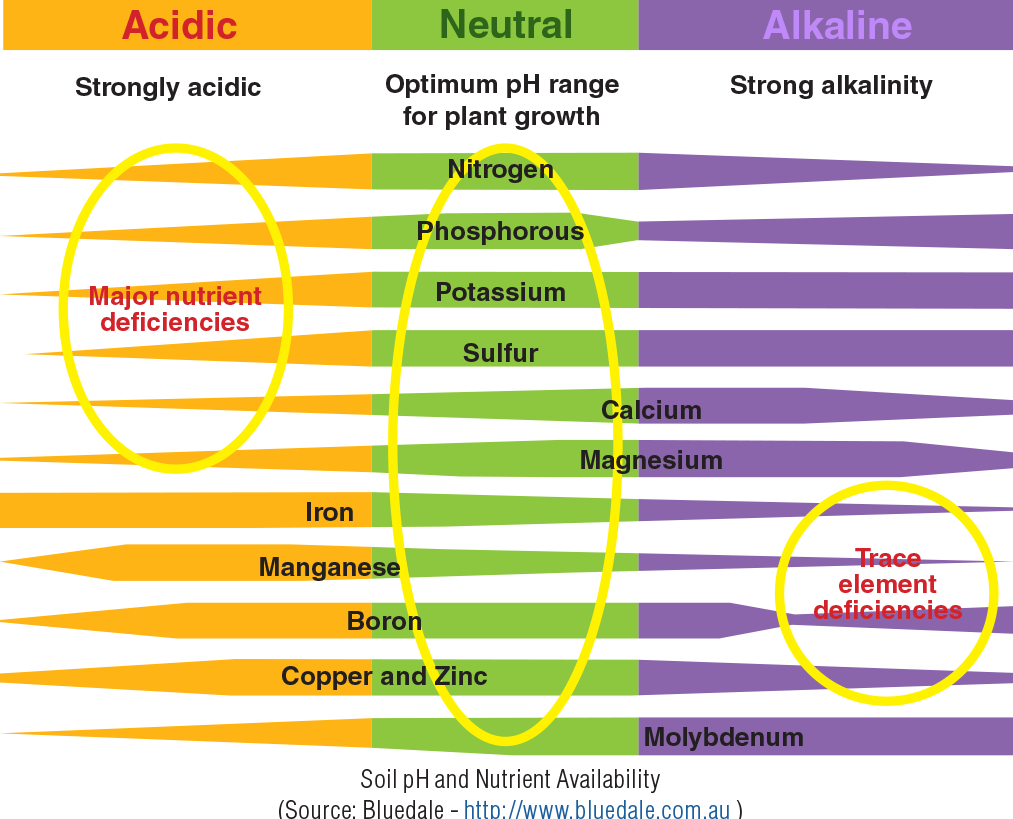
In most Australian soil tests, the pH of the soil is measured in water (pH(water)) or calcium chloride (pH CaCl2). Soil pH CaCl2 values are usually between 0.5 to 1.1 units lower than pH(water). The pH(water) value readily reflects current soil conditions, but is subject to seasonal variations. The CaCl2 test is useful for long-term monitoring of pH and is less subject to seasonal variations. Aim to keep the pH level above 5.3 (water) or 4.5 (CaCl2).
Plant-available nitrogen
It is difficult to measure the amount of nitrogen (N) available for plant growth in soils, because the form and availability of nitrogen in the soil can change quickly, particularly in improved pastures. Therefore, by the time the soil samples are received and analysed by the laboratory, the amount of mineral N in the sample may have changed.
Even if the amount of mineral N is correctly analysed by the laboratory, by the time the soil test results are returned to the farmer, changes may have already occurred in the N content of the soil.
The plant-available forms of nitrogen are ammonium-N (NH4-N) and nitrate-N (NO3-N). The abbreviation NH4-N means nitrogen in the ammonium form and the abbreviation NO3-N means nitrogen in the nitrate form.
Soil concentrations of NO3-N and NH4-N depend on biological activity and, therefore, fluctuate with changes in conditions, such as temperature and moisture. Nitrate is easily leached from the soil following high rainfall or excessive irrigation.
Soil tests can determine NO3-N and NH4-N concentrations at the time of sampling, but do not necessarily reflect future conditions.
Available phosphorus
Phosphorus (P) is essential for plant growth and is vital for early root formation. Soil minerals can react strongly with applied phosphorus and only a small proportion may be available for plant uptake.
Plant-available phosphorus can be tested using different methods, such Olsen, Bray or Colwell (depending on the region), and results are presented in milligrams per kilogram (or mg/kg) or parts per million (or ppm).
Available potassium
Potassium (K) is needed for a wide range of important processes within the plant, including cell wall development, flowering and seed set. Available potassium can be measured by different methods, such as Exchangeable K soil tests. Because the holding and supply capacity of potassium in soils can differ, the appropriate target for available potassium depends on soil type. When potassium levels are high, potassium inputs can be reduced from the fertiliser regime until levels fall.
Available sulphur
Sulphur is essential for nitrogen (N) fixation by legumes, such as lucerne or clovers. It is usually measured by the potassium chloride (KCl 40) test and is reported as mg/kg. This test takes into account some of the sulphur that will become available during the growing season from the breakdown of organic forms of sulphur.
Sulphur is considered adequate when the levels are > 4 mg/kg using the CPC test. Sulphur is considered adequate when the levels are > 8 mg/kg using the Blair (KCl 40) test. Plant analysis, especially a nitrogen-sulphur (N:S) ratio, is useful for diagnosing a sulphur deficiency.
Nutrients calcium, magnesium, sodium, aluminium and hydrogen are also typically recorded as plant-available values in the report, in addition to the extractable/exchangeable values.
Cation exchange capacity
The cation exchange capacity (CEC) of a soil is the measure of the soil’s capacity to hold important cations (positively charged ions), such as calcium, magnesium, sodium and potassium. Some laboratories also include aluminium.
The CEC measure provides an indication of the types and amount of calcium, potassium and magnesium available, and associated ratios, while exchangeable sodium is useful for determining potential soil structural problems. The CEC of the soil is largely dependent on the amount and type of clay and organic matter that is present (which results in greater CEC values than sandy soil).
The exchangeable cations are usually reported in unit of milli-equivalents per 100 grams (meq/100g) soil and also as a percentage of total cations.
Exchangeable calcium
Calcium is a necessary plant nutrient that plays a key role in maintaining soil structure and is generally present in high concentrations in the soil solution, even at low pH. Calcium deficiencies usually are found only on very acidic soils.
Exchangeable calcium should be > 5 meq/100g and in the range of 65-80% of the total cations present.
Exchangeable magnesium
Magnesium is also a necessary plant nutrient and is usually present in sufficient quantities to satisfy plant requirements. Exchangeable magnesium should be > 1.6 meq/100g and in the range of 10-20% of the total cations present. If exchangeable magnesium is more than 20% of the sum of cations present, it may result in potassium deficiency in plants and animals.
Calcium to magnesium ratio
Well-structured soils generally have twice the amount of exchangeable calcium to exchangeable magnesium. If the calcium to magnesium ratio is less than 2:1, this may indicate reduced soil stability.
In contrast, a calcium to magnesium ratio of more than 10:1 indicates a potential magnesium deficiency in plants and animals, including horses.
Exchangeable potassium
Potassium is an essential plant nutrient and is required in larger amounts. Exchangeable potassium should be > 0.5 meq/100g and in the range of 3-8% of the total cations present. If the exchangeable potassium level is more than 10% of the sum of cations, it may cause magnesium deficiency in plants and animals.
Magnesium to potassium ratio
The amount of magnesium should be one and a half times greater than the amount of potassium. If the ratio of magnesium to potassium is less than 1.5:1, this indicates an increased likelihood of magnesium deficiency in plants and potential grass tetany in classes of grazing animals.
Exchangeable sodium
Ideally, exchangeable sodium should be < 0.1 meq/100g and less than 1% of the total cations present.
If sodium makes up 6% or more of the total cations present, then the soil may be sodic and susceptible to dispersion – where a soil may lose structural integrity, compact and form surface crusts. The application of gypsum (CaSO4) can help alleviate excess sodium in the short term.
Exchangeable aluminium
High exchangeable aluminium concentrations can be common in very low pH soils and may be toxic to plants. High aluminium levels can be reduced by applying lime. Aluminium levels generally fall to harmless levels once the pH (water) exceeds 5.6-5.8. The exchangeable aluminium level should be less than 1% of the CEC.
Salinity (conductivity)
Soil salinity is a measure of the total soluble salts present. High levels of soluble salts in the root zone may affect water and nutrient uptake, and adversely affect plant growth. Plants are more susceptible to salinity in their germination and seedling stage than in later stages of growth.
Soil salinity is generally determined by measuring the electrical conductivity (EC) of the soil sample, with results decisiemens per metre (dS/m). Ideal levels are less than 0.2 dS/m.
Different pasture species have varying tolerance to soil salinity. Salt tolerance of plants is usually based on a different test – the electrical conductivity of a saturated extract method, ECe, which is also measured in dS/m. Salinity levels are satisfactory for all pasture species if the ECe is under 1 dS/m.
Summary

This article is meant to provide a basic understanding of how to read your soil tests and what some of these parameters tell you. Some soil reports may be more extensive and will report more values, such as trace minerals or heavy metals. This also depends on your chosen analysis package.
Additionally, the lab may record different measuring units. If this seems like too much information to digest at once, keep this article as a useful reference because I’ve aimed to bring the points that are more relevant to Australia and a horse property setting in particular.
Be sure to check the lab’s website, as they may provide additional information (or even services) than can help you with the interpretation of your results. You can always opt to get your soil test done and reported by a soil service or agronomist that, at the same time, may assist you with soil and pasture recommendations.
This article was published in the Horses and People Magazine May 2018
By Mariette van den Berg, PhD, BAppSc (Hons), RAnNutr
Mariette has a PhD in Equine Nutrition and Foraging Behaviour, is a RAnNutr equine nutritionist, a Certified Permaculture Designer and a dressage rider. She is the founder of MB Equine Services.